Dalia Savy
A
Anika P
AP Chemistry 🧪
269 resourcesSee Units
Resonance
Sometimes, there is not just one way to represent a molecule with a Lewis structure. If a structure can be drawn in multiple ways, the structure is known to have resonance. A good way of thinking of resonance is like mixing paint🎨.
When you draw out the two or three resonance structures of a molecule, those two/three structures make up the entire molecule. The actual structure is represented by an average of these 2-3 structures. This can lead to bond orders that are fractions, such as bond orders of 1/3 or 2/3.
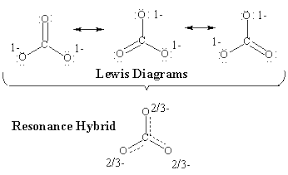
A common misconception is that structures that have resonance are truly able to exist with different bond connections. However, resonance is all about representing a molecule in varying ways just to make it clear that the molecule is actually an average of those representations in space. This will make more sense as we take a look at several examples.
How do you know when a structure has resonance?
Let's try drawing the lewis dot structure (LDS) of the polyatomic ion NO3-.
Recall some steps for this process that we discussed in the last study guide:
- Count the number of valence electrons that the molecule has in total. Nitrogen has 5 and an oxygen atom has 6. Since there are three oxygen atoms, we must account for each: 5+6+6+6 = 23. But...NO3- is a polyatomic ion and there is a charge attached to the molecule as a whole. The -1 charge indicates that there is one more electron, so there must be a total of 24 valence electrons represented in the LDS.
- Draw the central atom, which in this case, is nitrogen since there is only one nitrogen atom.
- Draw the 3 oxygen atoms surrounding nitrogen and three single bonds connecting them to nitrogen. Draw the full octets:
- Count the current number of valence electrons. There are 26, so somehow we have to get rid of 2 of them🤔. Remember, when there are too many electrons, we can replace a single bond and a lone pair with a double bond. In this case, if we change them all to double bonds, we won't have enough electrons! This is where resonance comes in: you only change one to a double bond.
When drawing the lewis structure of polyatomic ions, make sure you include the brackets and charge.
Now we count: we have 24! This is one way to draw the lewis dot structure of NO3-. How do we know which bond to make a double bond?
Simple answer: any of them since each of the three possibilities simultaneously exist in reality. There are three ways to draw this structure on paper:
Any of these would be acceptable. On the AP Exam itself, you would be required to draw all three side by side with these specific arrows between them ↔️.
In reality...🤔
In reality, the structure doesn't have 2 single bonds and 1 double bond. It has a 4/3 bond order. There is just no way to represent 4/3s of a bond.
Bond Order?
How did I know it was a 4/3s bond? The easiest way to know the bond order is ( # of lines / # of spaces ). There are 4 lines or bonds and 3 spaces for bonds. A 4/3s bond is between a single bond and a double bond. Therefore, it is stronger than a single bond but weaker than a double bond. A 4/3 bond is what is represented by these three structures.
As mentioned before, on the AP Exam, draw all possible structures side-by-side with double-sided arrows between each. You may also write the word "resonance" somewhere on the page to clarify what you know and break down the question for yourself.
👉Now try drawing the LDS of nitrite (another polyatomic ion). The answer is at the very end of the guide!
Formal Charge
Formal charge is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms. It reflects the electron count associated with the atom compared to the isolated neutral atom. It is used to predict the correct placement of electrons.
Here is an overall diagram, but let's get into the nitty-gritty.

How do you calculate formal charges?
The easiest way to calculate the formal charge of an atom is to do (# of valence electrons - # of dots - # of dashes). This might may not be the most technical but it's the easiest way to remember it!
Look at the image above at the SCN- ion to the left and the way they calculated formal charge:
- For the sulfur atom, they took the number of valence electrons (6) and subtracted it by the total number of lone electrons (2) and single bonds with carbon (3 [but there really is a triple bond]). This tells you that sulfur has a formal charge of +1 when drawn in SCN- with that representation of electrons.
When do I check the formal charge of an atom?
It is good to always check your calculation of formal charge since it is easy to miscount something. If anything, just check it if an element past element 14 is involved (because that is when you can break the octet rule).
Example with Formal Charge
Let's draw the LDS of phosphate (PO4-3):
- There should be 5+6+6+6+6+3 valence electrons, so 32 total.
- Draw Phosphorus in the center and 4 oxygen atoms surrounding it with single bonds and full octets:
- Looking at this lewis dot structure, we count 32 valence electrons and think it is perfect! However, phosphorus is element 15 so we should be looking at its formal charge to make sure the placement of electrons is correct.
- Calculating the formal charge:
- P: 5 valence electrons - 0 dots - 4 bonds = +1 formal charge
- O: 6 valence electrons - 6 dots - 1 bond = -1 formal charge
- When we are looking at the formal charges, we want the central atom to always have a formal charge of 0 or the most electronegative atom to have a negative formal charge. A formal charge of 0 means the electrons are localized, or not moving, and that representation of the molecule is the most stable.
- One way we can lower the formal charge of Phosphorus to 0 is by adding a double bond:
- Now let's check formal charge once more:
- P: 5 valence electrons - 5 bonds = 0 (Perfect🥳)
- Top 3 Oxygen atoms: 6 valence electrons - 6 dots - 1 bond = -1
- Botton oxygen atom: 6 valence electrons - 4 dots - 2 bonds = 0
Now, instead of having no atoms with a formal charge of 0, we have two atoms with a formal charge of 0. The electrons are placed correctly, resulting in a stable bond🎉.
The polyatomic ion also has resonance now, so you should draw all 4 ways of representing it and know the bonds are 5/4 order.
Quick Tip: An easy way to check if you calculated formal charge correctly is if the sum of the formal charges = the charge of the ion.
Since there were 3 oxygen atoms with a -1 charge, the sum is -3, which is equal to the -3 charge of the polyatomic ion.
AP FRQ Practice Questions
The following questions are pieces of free-response questions that were on previous AP Chemistry exams. These exams can be found on the College Board website.
AP Chemistry Exam 2016 - #2e
The HCO3− ion has three carbon-to-oxygen bonds. Two of the carbon-to-oxygen bonds have the same length and the third carbon-to-oxygen bond is longer than the other two. The hydrogen atom is bonded to one of the oxygen atoms. In the box below, draw a Lewis electron-dot diagram (or diagrams) for the HCO3 − ion that is (are) consistent with the given information.
They give you a large box for you to draw your answer in, but try this on your own!
AP Chemistry Exam 2017 - #1c
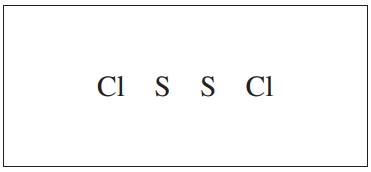
S2Cl2 is a product of the reaction. In the box below, complete the Lewis electron-dot diagram for the S2Cl2 molecule by drawing in all of the electron pairs.
AP Chemistry Exam 2017 #2a
Two possible Lewis electron-dot diagrams for fulminic acid, HCNO, are shown below. Explain why the diagram on the left is the better representation for the bonding in fulminic acid. Justify your choice based on formal charges.

2016 #2e Scoring Guidelines
You are given the ion and its charge and asked to draw the Lewis dot structure. Taking a look at HCO3-, you should calculate that there are a total of 23 valence electrons present, plus the additional electron from the negative charge: 24.
When you draw the structure out, you should place carbon in the center as the central atom. From there, always attach the multiple oxygen atoms to the central atom. If there is a hydrogen atom, you can basically bond it to any of the oxygens.
Once you have the atoms drawn and the full octets filled in, you count 26 valence electrons. We have too many electrons, so we must replace one single bond and lone pair with a double bond. Since there are two single bonds that you could convert to a double bond, this structure has resonance!
Here is an acceptable answer:
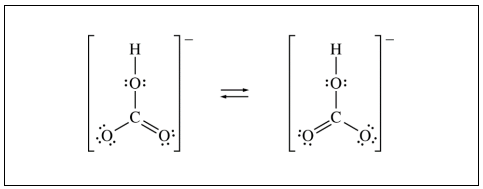
This is what College Board had on their site for full credit, but their arrow is the same as the double-headed arrow we discussed. Just please make sure you do not draw an equilibrium arrow by accident.
If one were to get this question correct, two full points would be earned:
- One point is given for drawing a correct Lewis structure with brackets and a charge.
- One point is given for indicating that there is resonance by drawing both structures with an arrow in between.
2017 #1c Scoring Guidelines
The following answer is correct and would earn you one full point:
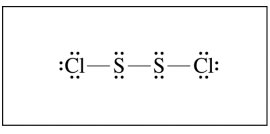
2017 #2a Scoring Guidelines
The basis of this question is calculating formal charges, and they even give you that hint! The best representation of a structure is one where the most electronegative atom has a negative formal charge.
This part was worth two points, one for the correct calculation of formal charge and one for a correct explanation of which is the better representation.
The following is a sample response:
- Point #1: In the diagram on the left, the C atom has a formal charge of zero and the O atom has a formal charge of -1. In the diagram on the right, the C atom has a formal charge of -1 and the O atom has a formal charge of zero.
- Point #2: The diagram on the left is the better representation because it puts the negative formal charge on oxygen, which is more electronegative than carbon.
Browse Study Guides By Unit
⚛️Unit 1 – Atomic Structure & Properties
🤓Unit 2 – Molecular & Ionic Bonding
🌀Unit 3 – Intermolecular Forces & Properties
🧪Unit 4 – Chemical Reactions
👟Unit 5 – Kinetics
🔥Unit 6 – Thermodynamics
⚖️Unit 7 – Equilibrium
🍊Unit 8 – Acids & Bases
🔋Unit 9 – Applications of Thermodynamics
🧐Exam Skills
📚Study Tools

Fiveable
Resources
© 2025 Fiveable Inc. All rights reserved.